In a major leap for medical technology, China’s National Medical Products Administration (NMPA) has approved the world’s first implantable brain-computer interface (BCI) for commercial use.
The device, developed by Shanghai-based Neuracle Medical Technology (NMT), is now legally allowed to be sold and used in hospitals to help people with severe paralysis regain control of their hands.
This approval, announced on March 13, 2026, marks the first time any country has cleared an invasive BCI (a device that sits on or inside the brain to read neural signals) for widespread clinical and commercial application.
Until now, similar technologies from companies like Neuralink (U.S.), Synchron (U.S./Australia), and Blackrock Neurotech (U.S.) have only been used in limited human trials or under special permissions, not as approved medical products available to doctors and patients.
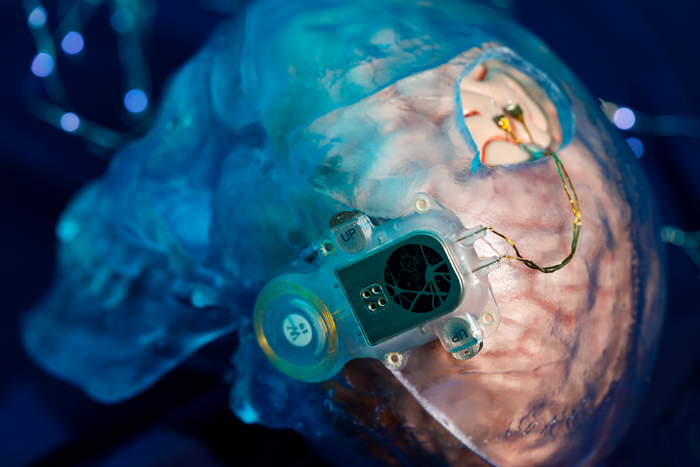
The Neuracle BCI, called the NEO system, is a small, coin-sized implant placed on the outer membrane of the brain (a minimally invasive procedure compared to deeper implants). It wirelessly reads brain signals related to intended hand movements.

These signals are sent to an external pneumatic robotic glove worn on the hand. When the patient thinks about grasping or moving, the glove inflates and deflates to help the fingers close or open, allowing actions like holding a cup, using a spoon, or pressing a button.
The system is designed for adults aged 18–60 who have quadriplegia (paralysis of all four limbs) caused by cervical spinal cord injuries at levels C2–C6 (graded A–C on the ASIA impairment scale). They must have been injured for at least one year and be medically stable.
In clinical trials involving 36 patients, the device successfully improved hand function and daily living activities without major safety issues reported.
Brain-computer interfaces have been studied for decades, but turning them into approved medical tools has taken time because of safety, reliability, and regulatory challenges.
The U.S. Food and Drug Administration (FDA) has approved some BCIs for limited human trials (Neuralink received clearance in 2023 and has implanted devices in a small number of patients), but none have yet reached full commercial status anywhere in the world.
China’s decision to approve Neuracle’s device first gives the country a clear lead in bringing invasive BCI technology to real patients.
The approval aligns with China’s national strategy to lead in future industries, including neurotechnology, robotics, and artificial intelligence. It also shows how quickly regulatory bodies can move when a device meets safety and effectiveness standards.
For patients with spinal cord injuries, this technology could restore basic hand movements sooner than waiting for deeper or more experimental implants.
Globally, it raises important questions: How will other countries respond? Will faster approvals in one nation push others to speed up their own reviews?
And how will this technology be made available and affordable in regions with high rates of spinal injuries, such as many parts of Africa, Asia, and Latin America, where road accidents and falls are common causes of paralysis?
While the Neuracle device is focused on hand function for paralysis, the approval opens the door for future BCIs to help with communication, mobility, or other conditions.
It also highlights the global race in neurotech: companies and governments are investing heavily because BCIs could one day treat paralysis, blindness, deafness, Parkinson’s, epilepsy, depression, and more.
At the same time, the technology brings ethical concerns (privacy of brain data, potential misuse, and long-term safety) that will need careful oversight. The Neuracle approval is a powerful signal that the era of commercial brain implants has begun, and the world is watching closely to see what comes next.
This story is developing. Expect more details as hospitals begin using the device and as other countries respond to China’s move. For now, it stands as a landmark moment: the first time an implantable brain-computer interface has crossed from research into approved medical practice.